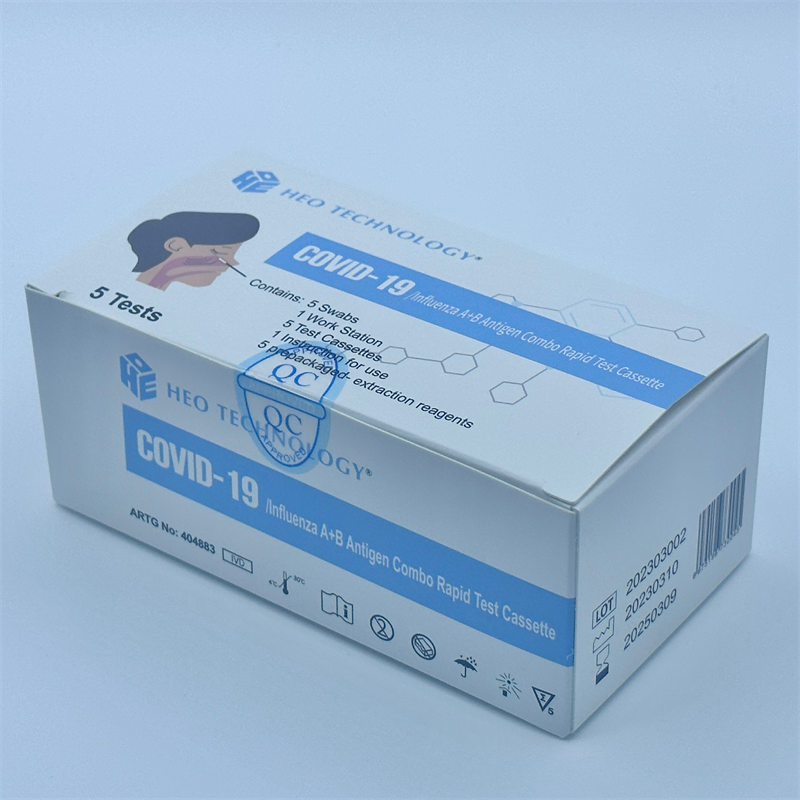
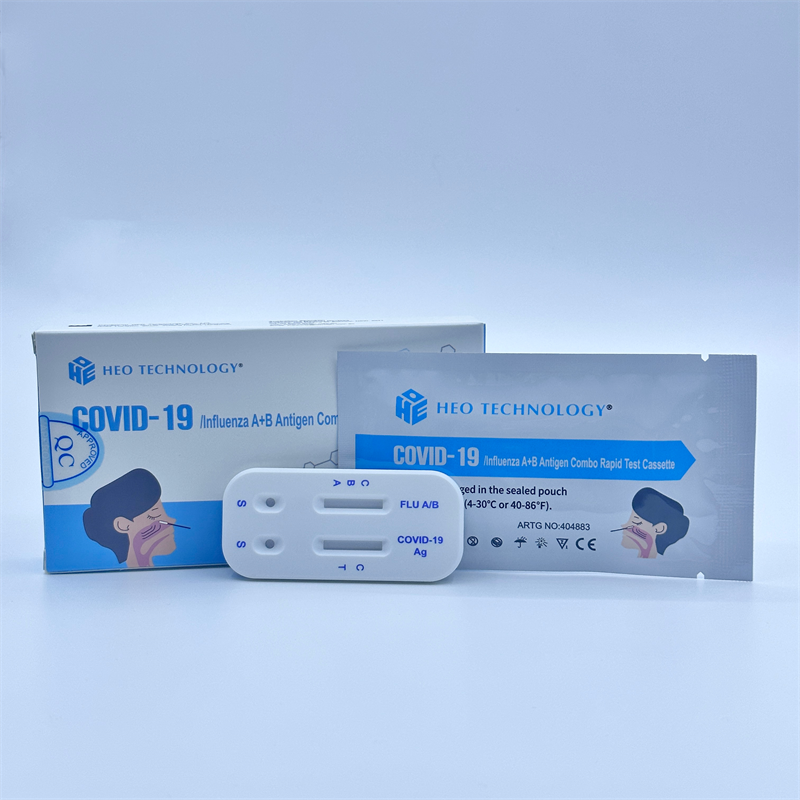
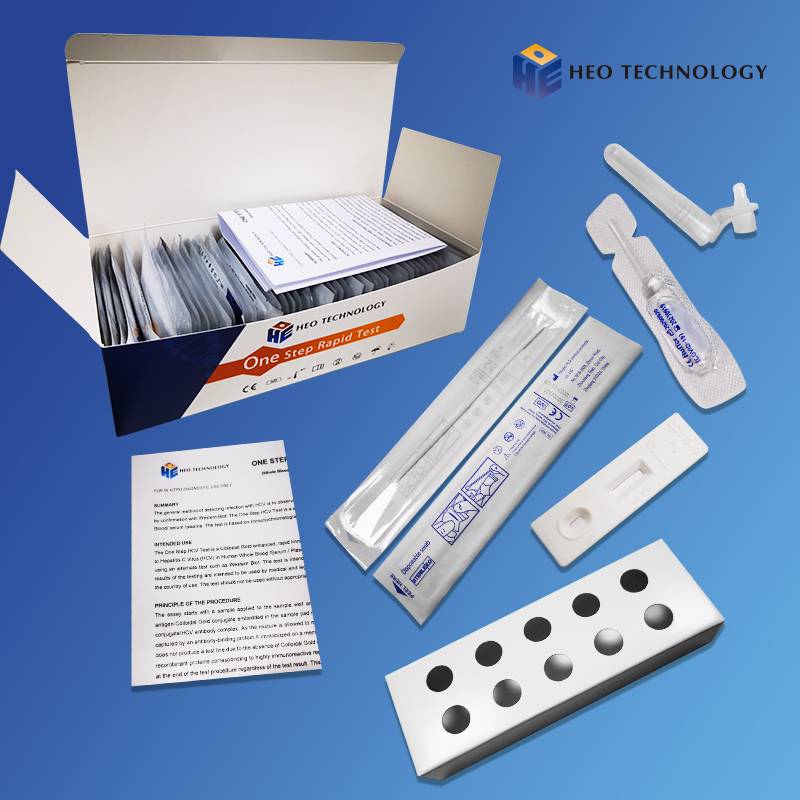
khoom
txog peb

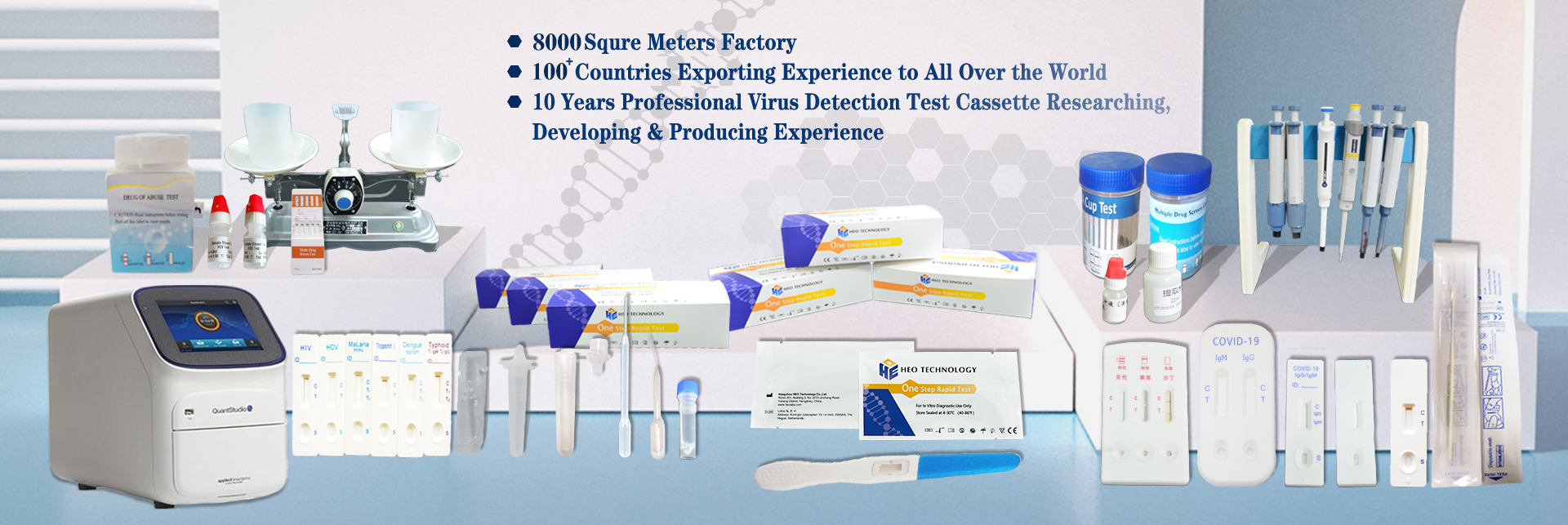
peb ua dab tsi
Hangzhou HEO Technology Co., LTD tau cog lus rau Kev Tshawb Fawb, Kev Tsim Kho thiab tsim tawm ntawm In-Vitro Diagnostic (IVD) Test Cassettes thiab Lwm Cov Khoom Siv Kho Mob rau 10 xyoo dhau los.
Peb Ua tau zoo tsim kev lag luam kev sib raug zoo nrog 60 ntau lub teb chaws thoob plaws ntiaj teb, xws li Southeast Asia, Europe, Latin American, South America, African lub teb chaws thiab lwm yam. Peb lub tuam txhab npog thaj tsam ntawm ntau tshaj 3000 square meters rhiav.Peb muaj ib qho kev sim cog lus pov thawj los ntawm National Food and Drug Administration thiab C-qib purification 1100 square meters rhiav.Txij li thaum nws tsim nyob rau xyoo 2011, peb tau pib tsom mus rau kev nyab xeeb ntawm cov zaub mov thiab In-Vitro Diagnostic Reagents 'kev tshawb fawb, kev tsim kho, thiab peb tau nruj me ntsis ua raws li ISO13485 thiab ISO9001 hauv kev tswj hwm zoo thiab tag nrho cov txheej txheem tsim khoom.
Peb cov ntawv xov xwm, cov ntaub ntawv tshiab tshaj plaws txog peb cov khoom, xov xwm thiab cov khoom tshwj xeeb.
Nyem rau phau ntawv
daim ntawv thov
-
 500
500 Muaj txiaj ntsig
-
 1000,0000
1000,0000 Tus nqi
-
 20
20 Kev koom tes
-
 24
24 sijhawm ua haujlwm
-
 35
35 Benchmark
xov xwm
Qhov Zoo Tshaj Plaws Ua Ntej Ua Ntej!